Abstract
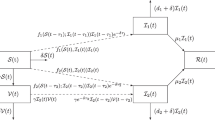
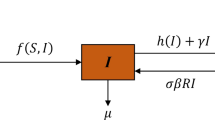
Multi-strain infectious diseases, which are usually prevented from spreading widely by vaccination, have two main transmission mechanisms: competitive exclusion and co-existence. In this paper, a stochastic multi-strain coinfection model with amplification and vaccination is developed. For the deterministic model, the basic reproduction number \(R_0\) and fixed points are provided. For the stochastic model, we first prove the existence and uniqueness of the positive solution under any initial value. Then, a portion of those infected with the common strain will always become patients with the amplified strain, which increases the risk of death from the disease. Therefore, we verified that patients with common strains would become extinct if \(R_1^s<1\). Furthermore, by constructing the Lyapunov function, we find that model (3) has a unique ergodic stationary distribution if \(R_0^S>1\). Particularly, we get a concrete form of the probability density of the distribution \(\kappa (\cdot )\) near equilibrium \(E^*\), where \(E^*\) is the quasi-local equilibrium of the stochastic model. Finally, the results are verified by numerical simulation. The results show that vaccination can control disease outbreaks or even eliminate them.








Similar content being viewed by others
References
Overview of the Epidemic of National Statutory Infectious Diseases in 2021. National Health Commission
Bonhoeffer, S., Nowak, M.A.: Mutation and the evolution of virulence. Proc. R. Soc. Lond. B. 258(1352), 133–140 (1994)
Martcheva, M.: A non-autonomous multi-strain SIS epidemic model. J. Biol. Dyn. 3(2–3), 235–251 (2009)
Kuddus, M.A., McBryde, E.S., Adekunle, A.I., White, L.J., Meehan, M.T.: Mathematical analysis of a two-strain disease model with amplification. Chaos Soliton Fract. 143, 110594 (2021) https://doi.org/10.1016/j.chaos.2020.110594
He, D.H., Ali, S.T., Fan, G.H., Gao, D.Z., Song, H.T., Lou, Y.J., Zhao, S., Cowling, B.J., Stone, L.: Evaluation of effectiveness of global COVID-19 vaccination campaign. Emerg. Infect. Dis. 28(9), 1873–1876 (2022)
Kaur, S.P., Gupta, V.: COVID-19 vaccine: a comprehensive status report. Virus Res. 288, 198114 (2020). https://doi.org/10.1016/j.virusres.2020.198114
Yavuz, M., Coşar, F.Ö., Günay, F., Özdemir, F.N.: A new mathematical modeling of the COVID-19 pandemic including the vaccination campaign. Open J. Model. Simul. 9(3), 299–321 (2021)
Wang, X.Y., Yang, J.Y., Han, Y.: Threshold dynamics of a chronological age and infection age structured cholera model with Neumann boundary condition. Z. Angew. Math. Phys. 74(4), 170 (2023)
Yang, J.Y., Yang, L., Jin, Z.: Optimal strategies of the age-specific vaccination and antiviral treatment against influenza. Chaos Soliton Fract. 168, 113199 (2023) https://doi.org/10.1016/j.chaos.2023.113199
Saha, S., Samanta, G., Nieto, J.J.: Epidemic model of COVID-19 outbreak by inducing behavioural response in population. Nonlinear Dyn. 102, 455–487 (2020)
Saha, S., Samanta, G., Nieto, J.J.: Impact of optimal vaccination and social distancing on COVID-19 pandemic. Math. Comput. Simul. 200, 285–314 (2022)
Wu, H., Zhang, L., Li, H.L., Teng, Z.D.: Stability analysis and optimal control on a multi-strain coinfection model with amplification and vaccination. Results Phys. 50, 106556 (2023) https://doi.org/10.1016/j.rinp.2023.106556
Khajanchi, S., Bera, S., Roy, T.K.: Mathematical analysis of the global dynamics of a HTLV-I infection model, considering the role of cytotoxic T-lymphocytes. Math. Comput. Simul. 180, 354–378 (2021) https://doi.org/10.1016/j.matcom.2020.09.009
Bera, S., Khajanchi, S., Roy, T.K.: Stability analysis of fuzzy HTLV-I infection model: a dynamic approach. J. Appl. Math. Comput. 69(1), 171–199 (2023)
Han, B.T., Jiang, D.Q., Zhou, B.Q., Hayat, T., Alsaedi, A.: Stationary distribution and probability density function of a stochastic SIRSI epidemic model with saturation incidence rate and logistic growth. Chaos Soliton Fract. 142, 110519 (2021). https://doi.org/10.1016/j.chaos.2020.110519
Omame, A., Abbas, M., Din, A.: Global asymptotic stability, extinction and ergodic stationary distribution in a stochastic model for dual variants of SARS-CoV-2. Math. Comput. Simulat. 204, 302–336 (2023) https://doi.org/10.1016/j.matcom.2022.08.012
Zhang, X.H., Jiang, D.Q., Alsaedi, A., Hayat, T.: Stationary distribution of stochastic SIS epidemic model with vaccination under regime switching. Appl. Math. Lett. 59, 87–93 (2016). https://doi.org/10.1016/j.aml.2016.03.010
Zhao, Y.N., Jiang, D.Q., O’Regan, D.: The extinction and persistence of the stochastic SIS epidemic model with vaccination. Physica A 392(20), 4916–4927 (2013)
Li, Z.M., Zhang, T.L., Li, X.Q.: Threshold dynamics of stochastic models with time delays: A case study for Yunnan, China. Electron Res. Arch. 29(1), 1661–1679 (2021)
Cai, Y.L., Kang, Y., Wang, W.M.: A stochastic SIRS epidemic model with nonlinear incidence rate. Appl. Math. Comput. 305, 221–240 (2017). https://doi.org/10.1016/j.amc.2017.02.003
Rao, F., Wang, W.M., Li, Z.B.: Stability analysis of an epidemic model with diffusion and stochastic perturbation. Commun. Nonlinear Sci. 17(6), 2551–2563 (2012)
Zhang, S.Q., Yuan, S.L., Zhang, T.H.: Dynamic analysis of a stochastic eco-epidemiological model with disease in predators. Stud. Appl. Math. 149(1), 5–42 (2022)
Silver, S.D., Driessche, P., Khajanchi, S.: A dynamic multistate and control model of the COVID-19 pandemic. J. Public Health, 1–14 (2023). https://doi.org/10.1007/s10389-023-02014-z
Mollah, S., Biswas, S., Khajanchi, S.: Impact of awareness program on diabetes mellitus described by fractional-order model solving by homotopy analysis method. Ric. Mat., 1–26 (2022). https://doi.org/10.1007/s11587-022-00707-3
Sarkar, K., Mondal, J., Khajanchi, S.: How do the contaminated environment influence the transmission dynamics of COVID-19 pandemic? Eur. Phys. J. Spec. Top. 231(18–20), 3697–3716 (2022)
Sardar, M., Khajanchi, S.: Is the Allee effect relevant to stochastic cancer model? J. Appl. Math. Comput. 68(4), 2293–2315 (2022)
Dwivedi, A., Keval, R., Khajanchi, S.: Modeling optimal vaccination strategy for dengue epidemic model: a case study of India. Phys. Scr. 97(8), 085214 (2022). https://doi.org/10.1088/1402-4896/ac807b
Saha, S., Dutta, P., Samanta, G.: Dynamical behavior of SIRS model incorporating government action and public response in presence of deterministic and fluctuating environments. Chaos Solit. Fract. 164, 112643 (2022)
Zhao, Y.N., Jiang, D.Q.: The threshold of a stochastic SIS epidemic model with vaccination. Appl. Math. Comput. 243, 718–727 (2014). https://doi.org/10.1016/j.amc.2014.05.124
Ma, Z.E., Zhou, Y.C., Li, C.Z.: Qualitative and Stability Methods for Ordinary Differential Equations. Science Press, Beijing (2015)
Hasminskii, R.: Stochastic Stability of Differential Equations. Sijthoff and Noordhoff. Alphen aan den Rijn, NetherBlands (1980)
Shi, Z.F., Jiang, D.Q., Zhang, X.H., Alsaedi, A.: A stochastic SEIRS rabies model with population dispersal: stationary distribution and probability density function. Appl. Math. Comput. 427, 127189 (2022) https://doi.org/10.1016/j.amc.2022.127189
Zuo, W.J., Jiang, D.Q.: Stationary distribution and periodic solution for stochastic predator-prey systems with nonlinear predator harvesting. Commun. Nonlinear Sci. 36, 65–80 (2016)
Zhang, G., Li, Z.M., Din, A.: A stochastic SIQR epidemic model with lévy jumps and three-time delays. Appl. Math. Comput. 431, 127329 (2022). https://doi.org/10.1016/j.amc.2022.127329
Zhao, S.N., Yuan, S.L., Zhang, T.H.: The impact of environmental fluctuations on a plankton model with toxin-producing phytoplankton and patchy agglomeration. Chaos Soliton Fract. 162, 112426 (2022). https://doi.org/10.1016/j.chaos.2022.112426
Zhang, S.Q., Yuan, S.L., Zhang, T.H.: A predator–prey model with different response functions to juvenile and adult prey in deterministic and stochastic environments. Appl. Math. Comput. 413, 126598 (2022) https://doi.org/10.1016/j.amc.2021.126598
Samantaa, G., Bera, S.P.: Analysis of a Chlamydia epidemic model with pulse vaccination strategy in a random environment. Nonlinear Anal-Model. 23(4), 457–474 (2018)
Nguyen, D.H., Yin, G., Zhu, C.: Long-term analysis of a stochastic SIRS model with general incidence rates. SIAM J. Appl. Math. 80(2), 814–838 (2020)
Liu, Q., Shi, Z.F.: Analysis of a stochastic HBV infection model with DNA-containing capsids and virions. J. Nonlinear Sci. 33(2), 23 (2023). https://doi.org/10.1007/s00332-022-09883-w
Zhou, B.Q., Jiang, D.Q., Dai, Y.C., Hayat, T.: Threshold dynamics and probability density function of a stochastic avian influenza epidemic model with nonlinear incidence rate and psychological effect. J. Nonlinear Sci. 33(2), 29 (2023)
Gardiner, C.W., et al.: Handbook of Stochastic Methods. Springer, Berlin (1985)
Roozen, H.: An asymptotic solution to a two-dimensional exit problem arising in population dynamics. SIAM J. Appl. Math. 49(6), 1793–1810 (1989)
Higham, D.J.: An algorithmic introduction to numerical simulation of stochastic differential equations. SIAM Rev. 43(3), 525–546 (2001)
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant Nos. 12271421, 12371504) and the Shaanxi Province Innovation Talent Promotion Plan Project (Grant No: 2023KJXX-056)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
For the same dimensional symmetric matrices \(Q_1\) and \(Q_2\), we have
\(Q_1\succ 0\): \(Q_1\) is a positive definite matrix,
\(Q_1\succeq 0\): \(Q_1\) is a positive semi-definite matrix,
\(Q_1\succeq Q_2\): \(Q_1-Q_2\) is at least a positive semi-definite matrix.
Moreover, for any invertible matrix Q, it is found that \(Q\Sigma Q^{T}\succ 0\) if \(\Sigma \succ 0\). Furthermore, define \(\Sigma _j\) as the solutions of the following algebraic equations
where
Consider the algebraic equation
which the related proof can be divided into two conditions:
Case 1. If \((\aleph _1)\) is satisfied, an intuitive calculation shows that
The characteristic polynomial of \(\mathcal {A}\) is that \(\varphi _\mathcal {A}(\lambda )=(\lambda -a_{11})(\lambda I_4-\mathcal {H}_1)\), where \(\mathcal {H}_1=(a_{ij})_{\{2\le i,j\le 4\}}\) is the matrix. Obviously, \(\mathcal {A}\) has one of the eigenvalue \(\lambda =a_{11}\). Based on the Theorem 2.1, we know that \(a_{11}>0\), which implies that \(\vartheta _{11}\succeq 0\) and
Case 2. If \((\hbar _1)\) is satisfied, \(a_{21}\ne 0\), or \(a_{31}\ne 0\), or \(a_{41}\ne 0\) can be obtained. We must illustrate that the elements \(a_{j1}~(j = 2, 3, 4,)\) have the equivalent status in \(\mathcal {A}\).
Define the invertible matrices \(\mathcal {J}_1\) and \(\mathcal {J}_2\) as follows
which satisfies \(\dot{\mathcal {A}}=\mathcal {J}_1\mathcal {A}\mathcal {J}_1^{-1}\), \(\ddot{\mathcal {A}}=\mathcal {J}_2\mathcal {A}\mathcal {J}_2^{-1}\), \(\dot{\Sigma _1}=\mathcal {J}_1\Sigma _1\mathcal {J}_1^T\) and \(\ddot{\Sigma _1}=\mathcal {J}_2\Sigma _1\mathcal {J}_2^T\).
Then (28) can be equivalently expressed as
Now, we can find that \(\Sigma _1\), \(\dot{\Sigma _1}\) and \(\ddot{\Sigma _1}\) not only have the same positive definiteness, but also they are Hurwitz matrix. Additionally, we can obtain that \(\dot{a_{21}}=a_{31}\) and \(\ddot{a_{21}}=a_{41}\). Therefore, we only consider the condition of \(a_{21}\ne 0\).
Let \(\mathcal {B}=\mathcal {J}_3\mathcal {A}\mathcal {J}_3^{-1}\), where the elimination matrix \(\mathcal {J}_3\) is given by
By calculation, we denote
There is \(b_{21}=a_{21}\ne 0\), equation (28) can be rewritten as:
Next, the parameters \((b_{32}, b_{42})\) are similarly considered for two conditions:
Case 2.1 If \((\aleph _2)\) is satisfied, by direct computation
where O is zero matrix and \(\mathcal {H}_2=(\varsigma _{ij})_{2\times 2}\) is the symmetric matric, where
It is easy to obtain the characteristic polynomial of \(\mathcal {A}\):
where \(\mathcal {H}_4=(b_{ij})_{\{3\le i,j\le 4\}}\) is the matrix. Because of the \(\mathcal {A}\) is a Hurwtiz matrix, all of the roots of the equation \(\lambda ^2-(b_{11}+b_{22})\lambda +(b_{11}b_{22}-b_{12}b_{21})=0\) have negative real part. Moreover, we have \(b_{11}+b_{22}<0\) and \(b_{11}b_{22}-b_{12}b_{21}>0\). Since \(b_{21}\ne 0\), we have
which shows that \(\mathcal {H}_2\succ 0\) and \(\mathcal {J}_3\Sigma _1\mathcal {J}_3^T\succeq 0\).
Define two positive semi-definite matrices \(\vartheta _{12}\) and \(\tilde{\vartheta _{12}}\) by
We have
Through \(\vartheta _{12}\succeq 0\), it can be obtained that \(\mathcal {J}_3^{-1}\tilde{\vartheta _{12}}(\mathcal {J}_3^{-1})^T\succeq 0\) and
Case 2.2 If \((\hbar _2)\) is satisfied, Similar to Case 2, we have \(b_{32}\ne 0\) or \(b_{42}\ne 0\) and the elements \(b_{m2}~(m=3,4)\) have the equivalent status in \(\mathcal {B}\). Therefore, we only consider the condition of \(b_{32}\ne 0\).
Define the invertible matrices \(\mathcal {J}_4\) as follows
which satisfies \(\hat{\mathcal {B}}=\mathcal {J}_4\mathcal {B}\mathcal {J}_4^{-1}\) and \(\hat{\Sigma _1}=\mathcal {J}_4\Sigma _1\mathcal {J}_4^T\). That is, (30) also can be rewritten as \(\mathcal {J}_4\mathcal {N}_1\mathcal {J}_4^T+\hat{\mathcal {B}}\hat{\Sigma _1}+\hat{\Sigma _1}\hat{\mathcal {B}}^T=0\).
Let \(\mathcal {C}=\mathcal {J}_5\mathcal {B}\mathcal {J}_5^{-1}\), where \(\mathcal {J}_2\) is called the second elimination metric
By calculation, it can be denoted
We have \(c_{21}=b_{21}=a_{21}(\ne 0)\) and \(c_{32}=b_{32}(\ne 0)\). Equations (30) or (28) can be rewritten as:
Next, the parameter \(c_{43}\) is similarly considered for two conditions:
Case 2.2.1 If \((\hbar _3)\) is satisfied, let \(\mathcal {D}=\mathcal {J}_6\mathcal {C}\mathcal {J}_6^T\), where the standardized transformation matrix
where
By calculation, it can be obtained
where \(\varphi _1, \varphi _2, \varphi _3, \varphi _4\) are the same as above. Furthermore, equation (32) can be rewritten as:
Moreover, using the Lemma 2.2, we can obtain that the matrix \(\Sigma _1\) is positive definite whose explicit form is
Compared the \(\mathcal {N}_1^2+\mathcal {D}\Sigma _1+\Sigma _1\mathcal {D}^T=0\), we have \(\Sigma _1=\Xi ^{-2}(\mathcal {J}_6\mathcal {J}_5\mathcal {J}_3)\Sigma _1(\mathcal {J}_6\mathcal {J}_5\mathcal {J}_3)^T\), where \(\Xi =d_{11}\sigma _1\). Thus, the matrix \(\Sigma _1=\Xi ^{-2}(\mathcal {J}_6\mathcal {J}_5\mathcal {J}_3)^{-1}\Sigma _1((\mathcal {J}_6\mathcal {J}_5\mathcal {J}_3)^{-1})^T\) is at least a semi-positive matrix.
Case 2.2.2 If \((\aleph _3)\) is satisfied, then the characteristic polynomial of \(\mathcal {C}\) is
where
Using Theorem 1 and \(\mathcal {A}\) is a Hurwitz matrix, the solution of equation \(\lambda ^3+\xi _1\lambda ^2+\xi _2\lambda +\xi _3=0\) have roots with all negative real components, that is
Let \(\mathcal {D}=\mathcal {J}_7\mathcal {C}\mathcal {J}_7^{-1}\) and using the Lemma 2.3, where the standardized transformation matrix \(\mathcal {J}_7\)
By calculation, we have
where
Then, the equation (32) can be rewritten as
where the \(\Sigma _1\) is shown as
Compared the \(\mathcal {N}_1^2+\mathcal {D}\tilde{\Sigma _1}+\tilde{\Sigma _1}\mathcal {D}^T=0\), we have \(\tilde{\Sigma _1}=\Xi ^{-2}(\mathcal {J}_7\mathcal {J}_5\mathcal {J}_3)\Sigma _1 (\mathcal {J}_7\mathcal {J}_5\mathcal {J}_3)^T\), where \(\Xi =c_{21}c_{32}\sigma _1\). Thus, the matrix \(\Sigma _1=\Xi ^{-2}(\mathcal {J}_7\mathcal {J}_5\mathcal {J}_3)^{-1}\tilde{\Sigma _1}((\mathcal {J}_7 \mathcal {J}_5\mathcal {J}_3)^{-1})^T\) is at least a semi-positive matrix.
For \(\mathcal {N}_i~(i=2,3,4)\), its approach is similar to that of \(\mathcal {N}_1\). Therefore, we ignore it here. The proof is complete.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Niu, L., Chen, Q. & Teng, Z. Threshold Dynamics and Probability Density Function of a Stochastic Multi-Strain Coinfection Model with Amplification and Vaccination. Qual. Theory Dyn. Syst. 23, 94 (2024). https://doi.org/10.1007/s12346-024-00957-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12346-024-00957-6