Abstract
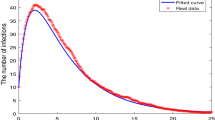
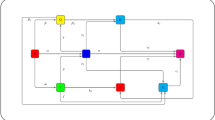
There have been reports of influenza virus resistance in the past, and because this virus has the potential of resistance to cause several pandemics and also is lethal, we investigate the conditions under which the strains coexist as a result. The non-resistant strain undergoes mutation, giving rise to the resistant strain. The incidence rates of the non-resistant and saturated-resistant strains are bi-linear and saturated, respectively. In this study, two flu strain models (resistant and non-resistant) are investigated in a fractal–fractional sense, and the presence of solutions, stability, and numerical simulations are examined for various orders and derivative dimensions. Using numerical values from freely accessible open resources, a numerical technique that is based on Lagrange’s interpolation polynomial is constructed and validated for a particular example.



Similar content being viewed by others
Availability of Data and Materials
No data were generated or analyzed during the current study.
References
Mohler, L., Flockerzi, D., Sann, H., Reichl, U.: Mathematical model of influenza A virus production in large-scale microcarrier culture. Biotechnol. Bioeng. 90, 46–58 (2005). https://doi.org/10.1002/bit.20363
Kreijtz, J.H.C.M., Bodewes, R., van Amerongen, G., Kuiken, T., Fouchier, R.A.M., Osterhaus, A.D.M.E., Rimmelzwaan, G.F.: Primary influenza A virus infection induces cross-protective immunity against a lethal infection with a heterosubtypic virus strain in mice. Vaccine 25, 612–620 (2007). https://doi.org/10.1016/j.vaccine.2006.08.036
Webster, R.G., Peiris, M., Chen, H., Guan, Y.: H5N1 outbreaks and enzootic influenza. Emerg. Infect. Dis. 12, 3–8 (2006). https://doi.org/10.3201/eid1201.051024
Rasmussen, S.A., Jamieson, D.J., Uyeki, T.M.: Effects of influenza on pregnant women and infants. Am. J. Obstet. Gynecol. 207, S3-8 (2012). https://doi.org/10.1016/j.ajog.2012.06.068
Bouvier, N.M., Lowen, A.C., Palese, P.: Oseltamivir-resistant influenza A viruses are transmitted efficiently among guinea pigs by direct contact but not by aerosol. J. Virol. 82, 10052–10058 (2008). https://doi.org/10.1128/JVI.01226-08
Ward, P., Small, I., Smith, J., Suter, P., Dutkowski, R.: Oseltamivir (Tamiflu) and its potential for use in the event of an influenza pandemic. J. Antimicrob. Chemother. 55, i5–i21 (2005). https://doi.org/10.1093/jac/dki018
Schunemann, H.J., Hill, S.R., Kakad, M., Bellamy, R., Uyeki, T.M., et al.: WHO Rapid Advice Guidelines for pharmacological management of sporadic human infection with avian influenza A (H5N1) virus. Lancet Infect. Dis. 7, 21–31 (2007). https://doi.org/10.1016/S1473-3099(06)70684-3
Baranovich, T., Saito, R., Suzuki, Y., Zaraket, H., Dapat, C., et al.: Emergence of H274Y oseltamivir-resistant A (H1N1) influenza viruses in Japan during the 2008–2009 season. J. Clin. Virol. 47, 23–28 (2010). https://doi.org/10.1016/j.jcv.2009.11.003
Monto, A.S., McKimm-Breschkin, J.L., Macken, C., Hampson, A.W., Hay, A., et al.: Detection of influenza viruses resistant to neuraminidase inhibitors in global surveillance during the first 3 years of their use. Antimicrob. Agents Chemotherap. 50, 2395–2402 (2006). https://doi.org/10.1128/AAC.01339-05
Carr, J., Ives, J., Kelly, L., Lambkin, R., Oxford, J., Mendel, D., Tai, L., Roberts, N.: Influenza virus carrying neuraminidase with reduced sensitivity to oseltamivir carboxylate has altered properties in vitro and is compromised for infectivity and replicative ability in vivo. Antivir. Res. 54, 79–88 (2002). https://doi.org/10.1016/s0166-3542(01)00215-7
Herlocher, M.L., Truscon, R., Elias, S., Yen, H.L., Roberts, N.A., et al.: Influenza viruses resistant to the antiviral drug oseltamivir: transmission studies in ferrets. J. Infect. Dis. 190, 1627–1630 (2004). https://doi.org/10.1086/424572
Abed, Y., Goyette, N., Boivin, G.: A reverse genetics study of resistance to neuraminidase inhibitors in an influenza A/H1N1 virus. Antivir. Ther. 9, 577–581 (2004)
Rameix-Welti, M.A., Enouf, V., Cuvelier, F., Jeannin, P., van der Werf, S.: Enzymatic properties of the neuraminidase of seasonal H1N1 influenza viruses provide insights for the emergence of natural resistance to oseltamivir. PLoS Pathogens 4, e1000103 (2008). https://doi.org/10.1371/journal.ppat.1000103
Baz, M., Abed, Y., Simon, P., Hamelin, M.E., Boivin, G.: Effect of the neuraminidase mutation H274Y conferring resistance to oseltamivir on the replicative capacity and virulence of old and recent human influenza A (H1N1) viruses. J. Infect. Dis. 201, 740–745 (2010). https://doi.org/10.1086/650464
Matsuzaki, Y., Mizuta, K., Aoki, Y., Suto, A., Abiko, C., et al.: A two-year survey of the oseltamivir-resistant influenza A (H1N1) virus in Yamagata, Japan and the clinical effectiveness of oseltamivir and zanamivir. Virol. J. 7(53), 1–8 (2010). https://doi.org/10.1186/1743-422X-7-53
Bloom, J.D., Gong, L.I., Baltimore, D.: Permissive secondary mutations enable the evolution of influenza oseltamivir resistance. Science 328, 1272–1275 (2010). https://doi.org/10.1126/science.1187816
Kaymakamzade, B., Baba, I.A., Hincal, E.: Global stability analysis of oseltamivir-resistant influenza virus model. Procedia Comput. Sci. 102, 333–341 (2016). https://doi.org/10.1016/j.procs.2016.09.409
Ives, J.A.L., Carr, J.A., Mendel, D.B., Tai, C.Y., Lambkin, R., et al.: The H274Y mutation in the influenza A/H1N1 neuraminidase active site following oseltamivir phosphate treatment leave virus severely compromised both in vitro and in vivo. Antivir. Res. 55, 307–317 (2002). https://doi.org/10.1016/s0166-3542(02)00053-0
Wu, Y., Ahmad, S., Ullah, A., Shah, K.: Study of the fractional-order HIV-1 infection model with uncertainty in initial data. Math. Probl. Eng. 2022(7286460), 1–16 (2022). https://doi.org/10.1155/2022/7286460
Ali, A., Alshammari, F.S., Islam, S., Khan, M.A., Ullah, S.: Modeling and analysis of the dynamics of novel coronavirus (COVID-19) with Caputo fractional derivative. Results Phys. 20, 103669 (2021). https://doi.org/10.1016/j.rinp.2020.103669
Ali, A., Islam, S., Khan, M.R., Rasheed, S., Allehiany, F.M., Baili, J., Khan, M.A., Ahmad, H.: Dynamics of a fractional order Zika virus model with mutant. Alex. Eng. J. 61(6), 4821–36 (2022). https://doi.org/10.1016/j.aej.2021.10.031
Ali, A., Hamou, A.A., Islam, S., Muhammad, T., Khan, A.: A memory effect model to predict COVID-19: analysis and simulation. Comput. Methods Biomech. Biomed. Eng. 26(5), 612–28 (2023). https://doi.org/10.1080/10255842.2022.2081503
Ali, A., Ullah, S., Khan, M.A.: The impact of vaccination on the modeling of COVID-19 dynamics: a fractional order model. Nonlinear Dyn. 110(4), 3921–40 (2022). https://doi.org/10.1007/s11071-022-07798-5
Aba Oud, M.A., Ali, A., Alrabaiah, H., Ullah, S., Khan, M.A., Islam, S.S.: A fractional order mathematical model for COVID-19 dynamics with quarantine, isolation, and environmental viral load. Adv. Differ. Equ. 2021(1), 1–9 (2021). https://doi.org/10.1186/s13662-021-03265-4
Ali, A., Iqbal, Q., Asamoah, J.K., Islam, S.: Mathematical modeling for the transmission potential of Zika virus with optimal control strategies. Eur. Phys. J. Plus 137(1), 146 (2022). https://doi.org/10.1140/epjp/s13360-022-02368-5
Ali, A., Iqbal, Q., Asamoah, J.K., Islam, S.: Mathematical modeling for the transmission potential of Zika virus with optimal control strategies. Eur. Phys. J. Plus 137(1), 46 (2022). https://doi.org/10.1140/epjp/s13360-022-02368-5
Butt, A.I., Rafiq, M., Ahmad, W., Ahmad, N.: Implementation of computationally efficient numerical approach to analyze a Covid-19 pandemic model. Alex. Eng. J. 69, 341–62 (2023)
Hanif, A., Kashif Butt, A.I., Ahmad, W.: Numerical approach to solve Caputo–Fabrizio-fractional model of corona pandemic with optimal control design and analysis. Math. Methods Appl. Sci. (2023). https://doi.org/10.1002/mma.9085
Ahmad, W., Abbas, M.: Effect of quarantine on transmission dynamics of Ebola virus epidemic: a mathematical analysis. Eur. Phys. J. Plus 136(4), 1–33 (2021)
Ahmad, W., Rafiq, M., Abbas, M.: Mathematical analysis to control the spread of Ebola virus epidemic through voluntary vaccination. Eur. Phys. J. Plus 135(10), 775 (2020). https://doi.org/10.1140/epjp/s13360-020-00683-3
Rafiq, M., Ahmad, W., Abbas, M., Baleanu, D.: A reliable and competitive mathematical analysis of Ebola epidemic model. Adv. Differ. Equ. 2020, 1–24 (2020). https://doi.org/10.1186/s13662-020-02994-2
Padder, A., Almutairi, L., Qureshi, S., Soomro, A., Afroz, A., Hincal, E., Tassaddiq, A.: Dynamical analysis of generalized tumor model with Caputo fractional-order derivative. Fractal Fract. 7(3), 258 (2023)
Qureshi, S., Abro, K.A., Gomez-Aguilar, J.F.: On the numerical study of fractional and non-fractional model of nonlinear Duffing oscillator: a comparison of integer and non-integer order approaches. Int. J. Model. Simul. 43(4), 362–75 (2023)
Jan, R., Qureshi, S., Boulaaras, S., Pham, V.T., Hincal, E., Guefaifia, R.: Optimization of the fractional-order parameter with the error analysis for human immunodeficiency virus under Caputo operator. Discrete Contin. Dyn. Syst. S (2023). https://doi.org/10.3934/dcdss.2023010
Qureshi, S., Akanbi, M.A., Shaikh, A.A., Wusu, A.S., Ogunlaran, O.M., Mahmoud, W., Osman, M.S.: A new adaptive nonlinear numerical method for singular and stiff differential problems. Alex. Eng. J. 74, 585–97 (2023)
Alquran, M., Ali, M., Gharaibeh, F., Qureshi, S.: Novel investigations of dual-wave solutions to the Kadomtsev–Petviashvili model involving second-order temporal and spatial–temporal dispersion terms. Partial Differ. Equ. Appl. Math. 8, 100543 (2023)
Atangana, A.: Fractal–fractional differentiation and integration: connecting fractal calculus and fractional calculus to predict complex system. Chaos Solitons Fractals 102, 396–406 (2017). https://doi.org/10.1016/j.chaos.2017.04.027
Khan, H., Ahmad, F., Tunc, O., Idrees, M.: On fractal–fractional Covid-19 mathematical model. Chaos Solitons Fractals 157, 111937 (2022). https://doi.org/10.1016/j.chaos.2022.111937
Gomez-Aguilar, J.F., Cordova-Fraga, T., Abdeljawad, T., Khan, A., Khan, H.: Analysis of fractal–fractional malaria transmission model. Fractals 28, 2040041 (2020). https://doi.org/10.1142/S0218348X20400411
Ghori, M.B., Naik, P.A., Zu, J., Eskandari, Z., Naik, M.U.: Global dynamics and bifurcation analysis of a fractional-order SEIR epidemic model with saturation incidence rate. Math. Methods Appl. Sci. 45(7), 3665–88 (2022)
Naik, P.A., Zu, J., Naik, M.U.: Stability analysis of a fractional-order cancer model with chaotic dynamics. Int. J. Biomath. 14(06), 2150046 (2021)
Naik, P.A., Ghoreishi, M., Zu, J.: Approximate solution of a nonlinear fractional order HIV model using Homotopy analysis method. Int. J. Numer. Anal. Model. 19(1) (2022)
Ahmad, A., Farman, M., Naik, P.A., Zafar, N., Akgul, A., Saleem, M.U.: Modeling and numerical investigation of fractional-order bovine babesiosis disease. Numer. Methods Partial Differ. Equ. 37(3), 1946–64 (2021)
Naik, P.A., Yavuz, M., Qureshi, S., Zu, J., Townley, S.: Modeling and analysis of COVID-19 epidemics with treatment in fractional derivatives using real data from Pakistan. Eur. Phys. J. Plus 135, 1–42 (2020)
Shah, K., Arfan, M., Mahariq, I., Ahmadian, A., Salahshour, S., Ferrara, M.: Fractal–fractional mathematical model addressing the situation of Corona virus in Pakistan. Res. Phys. 19, 103560 (2020). https://doi.org/10.1016/j.rinp.2020.103560
Ali, Z., Rabiei, F., Shah, K., Khodadadi, T.: Qualitative analysis of fractal–fractional order COVID-19 mathematical model with case study of Wuhan. Alex. Eng. J. 60, 477–489 (2021). https://doi.org/10.1016/j.aej.2020.09.020
Alqhtani, M., Saad, K.M.: Fractal–fractional Michaelis–Menten enzymatic reaction model via different kernels. Fractal Fract. 6, 13 (2022). https://doi.org/10.3390/fractalfract6010013
Saad, K.M., Alqhtani, M., Gomez-Aguilar, J.F.: Fractal–fractional study of the hepatitis C virus infection model. Res. Phys. 19, 103555 (2020). https://doi.org/10.1016/j.rinp.2020.103555
Etemad, S., Avci, I., Kumar, P., Baleanu, D., Rezapour, S.: Some novel mathematical analysis on the fractal–fractional model of the AH1N1/09 virus and its generalized Caputo-type version. Chaos Solitons Fractals 162, 112511 (2022). https://doi.org/10.1016/j.chaos.2022.112511
Rvachev, L.A., Longini, J.I.M.: A mathematical model for the global spread of influenza. Math. Biosci. 75(1), 3–22 (1985)
Beauchemin, C.A., Handel, A.: A review of mathematical models of influenza A infections within a host or cell culture: lessons learned and challenges ahead. BMC Public Health 11(1), 1–5 (2011)
Boianelli, A., Nguyen, V.K., Ebensen, T., Schulze, K., Wilk, E., Sharma, N., Stegemann-Koniszewski, S., Bruder, D., Toapanta, F.R., Guzman, C.A., Meyer-Hermann, M.: Modeling influenza virus infection: a roadmap for influenza research. Viruses 7(10), 5274–304 (2015)
Ain, Q.T., Khan, A., Abdeljawad, T., Gómez-Aguilar, J.F., Riaz, S.: Dynamical study of varicella-zoster virus model in sense of Mittag–Leffler kernel. Int. J. Biomath. 17, 2350027 (2023)
Khan, Z.A., Khan, A., Abdeljawad, T., Khan, H.: Computational analysis of fractional order imperfect testing infection disease model. Fractals 30(05), 2240169 (2022)
Thirthar, A.A., Abboubakar, H., Khan, A., Abdeljawad, T.: Mathematical modeling of the COVID-19 epidemic with fear impact. AIMS Math. 8(3), 6447–65 (2023)
Ain, Q., Khan, A., Ullah, M.I., Alqudah, M.A., Abdeljawad, T.: On fractional impulsive system for methanol detoxification in human body. Chaos Solitons Fractals 160, 112235 (2022)
Abro, K.A., Atangana, A.: Numerical and mathematical analysis of induction motor by means of AB-fractal–fractional differentiation actuated by drilling system. Numer. Methods Partial Differ. Equ. 38, 293–307 (2022). https://doi.org/10.1002/num.22618
Baba, I.A., Ahmad, H., Alsulami, M.D., Abualnaja, K.M., Altanji, M.: A mathematical model to study resistance and non-resistance strains of influenza. Res. Phys. 26, 104390 (2021). https://doi.org/10.1016/j.rinp.2021.104390
Veeresha, P., Ilhan, E., Prakasha, D.G., Baskonus, H.M., Gao, W.: A new numerical investigation of fractional order susceptible-infected-recovered epidemic model of childhood disease. Alex. Eng. J. 61(2), 1747–56 (2022)
Baishya, C., Achar, S.J., Veeresha, P., Kumar, D.: Dynamical analysis of fractional yellow fever virus model with efficient numerical approach. J. Comput. Anal. Appl. 140 (2023)
Gao, W., Veeresha, P., Cattani, C., Baishya, C., Baskonus, H.M.: Modified predictor-corrector method for the numerical solution of a fractional-order SIR model with 2019-nCoV. Fractal Fract. 6(2), 92 (2022)
Rahman, M.: Generalized fractal–fractional order problems under non-singular Mittag–Leffler kernel. Results Phys. 35, 105346 (2022)
Shah, K., Abdalla, B., Abdeljawad, T., Gul, R.: Analysis of multipoint impulsive problem of fractional-order differential equations. Bound. Value Probl. 2023(1), 1–7 (2023)
Li, B., Zhang, T., Zhang, C.: Investigation of financial bubble mathematical model under fractal–fractional Caputo derivative. Fractals 31(05), 1–3 (2023)
Ahmad, S., Shah, K., Abdeljawad, T., Abdalla, B.: On the approximation of fractal–fractional differential equations using numerical inverse Laplace transform methods. CMES Comput. Model. Eng. Sci. 135(3) (2023)
Atangana, A., Araz, S.I.: Mathematical model of COVID-19 spread in Turkey and South Africa: theory, methods, and applications. Adv. Differ. Equ. 2020(1), 1–89 (2020)
Araz, S.I.: Analysis of a Covid-19 model: optimal control, stability and simulations. Alex. Eng. J. 60(1), 647–658 (2021)
Atangana, A., Araz, S.I.: Modeling and forecasting the spread of COVID-19 with stochastic and deterministic approaches: Africa and Europe. Adv. Differ. Equ. 2021, 1–07 (2021)
Acknowledgements
H.K and J.A express their sincere thanks to Prince Sultan University and OSTİM Technical University for their endless support. The S.E and S.R would like to thank Azarbaijan Shahid Madani University.
Funding
No funds were received.
Author information
Authors and Affiliations
Contributions
The authors declare that the study was realized in collaboration with equal responsibility. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khan, H., Rajpar, A.H., Alzabut, J. et al. On a Fractal–Fractional-Based Modeling for Influenza and Its Analytical Results. Qual. Theory Dyn. Syst. 23, 70 (2024). https://doi.org/10.1007/s12346-023-00918-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12346-023-00918-5
Keywords
- Fractal–fractional operators
- Influenza mathematical modeling
- Hyers–Ulam-stability
- Interpolation
- Fractal dimension.